How to Modify Study Info
Certain patient information can be accessed and modified if needed. This can be done by following these steps:
Note- If a study has been interpreted and signed, the DOB and gender cannot be modified. This information is used in the diagnostic process and changing this could directly impact the treatment considerations and final diagnosis.
Note- If a study has been interpreted and signed, the DOB and gender cannot be modified. This information is used in the diagnostic process and changing this could directly impact the treatment considerations and final diagnosis.
- Access the “Pending Studies Queue” menu by going to “Home
Sleep Testing” > “Pending Studies Queue”
- Enter your search
criteria and click the "Search" button to locate the patient
- Click on
next to the patient's name to access the context menu
- Select "Modify
Study Info"
- Make the necessary correction.
- You also must include a
reason for the Edit to save the changes.
- After clicking "Save", a new report will be generated for you to view the changes.
There is not an option to Undo your changes once they have been saved, so please be sure to make necessary notes before modification.
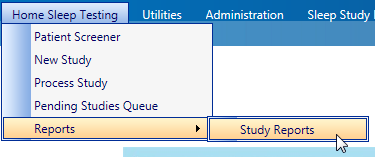
- Access the "Study Reports" menu by going to "Home Sleep Testing" > "Reports" > "Study Reports"
- Enter your search criteria and click the "Search" button
- Click on
next to the patient's name to access the context menu
- Select "Modify Study Info"
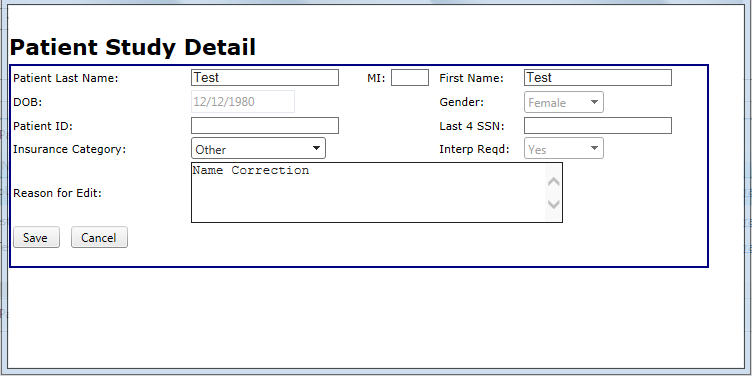
- If the study has been interpreted, certain fields will be "grayed" out and cannot be edited
- Make the necessary correction.
- You must include a reason for any modification in order to save the changes
- After clicking "Save", a new report will be generated for you to view the changes.
Related Articles
How To Initialize your ARES (PDF)
The ARES Device should be Initialized prior to each patient testing. This can be done by following these steps: Access the portal portal.watermarkmedical.com using Web browser Internet Explorer or Google Chrome Plug in your ARES device to the PC ...How To Upload your ARES (PDF)
The ARES Device should be uploaded after the patient has completed testing. This can be done by following these steps: Access the portal portal.watermarkmedical.com using Web browser Internet Explorer or Google Chrome Plug in your ARES device to the ...Customer FAQ's
Here are some commonly asked customer questions. If you do not see your question posted, please call customer support for further assistance at 877-710-6999. How old should you be to be to test with the ARES Device? The ARES Device is FDA approved ...How to Re-Interpret My Study
If a study has already been interpreted (signed) and released by the Interpreting physician but they need to make additional edits or changes please follow the steps below: Note: These steps must be performed by the Interpreting physician. This is ...Uploading the ARES 610 (Video)